
Insights+: EMA Marketing Authorization of New Drugs in April 2023
Shots:
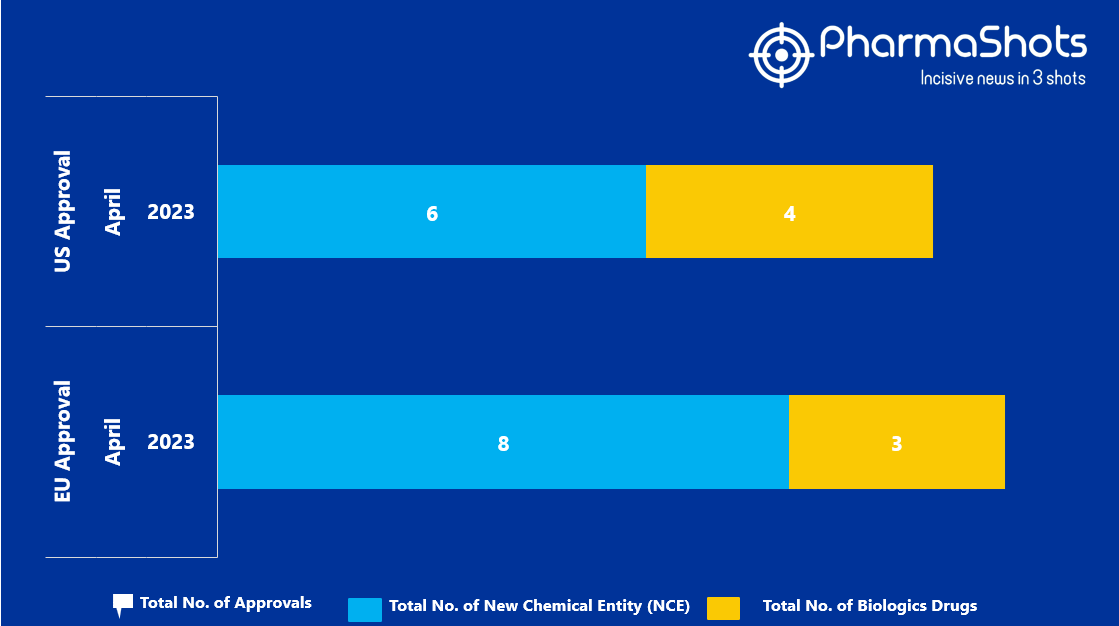
- The EMA approved 8 New Chemical Entity (NCE) and 3 Biologic Drugs in April 2023, leading to treatments for patients and advances in the healthcare industry
- In April 2023, the major highlights drugs were Rinvoq’s Approval for active Crohn's Disease, Opzelura for non-segmental vitiligo with facial involvement in adults and adolescents
- PharmaShots has compiled a list of a total of 11 new drugs approved by the EMA in April 2023
Breyanzi
Active ingredient: lisocabtagene maraleucel Approved: April 01, 2023
Company: BMS Disease: Large B-cell Lymphoma
- The EMA’s CHMP has recommended the approval of Breyanzi for adult patients with DLBCL, HGBCL, PMBCL & FL3B who relapsed within 12mos. from completion of, or are refractory to 1L chemoimmunotherapy
- The opinion was based on the P-III study (TRANSFORM) evaluating Breyanzi vs SoC consisting of salvage CT, followed by high-dose CT + HSCT in 184 patients. The EC’s final decision is expected within ~2mos. following receipt of the CHMP opinion
- The EC’s decision will be valid to all EU member states, Iceland, Norway, and Liechtenstein. Breyanzi is a CD19-directed CAR T cell therapy with a 4-1BB costimulatory domain & was approved in Japan for 2L treatment of r/r LBCL & in Japan, EU, Switzerland & Canada for r/r LBCL
Ultomiris
Active ingredient: ravulizumab Approved: April 03, 2023
Company: AstraZeneca Disease: Neuromyelitis Optica Spectrum Disorder
- The EMA’s CHMP has recommended Ultomiris for marketing authorization in the EU for adult patients with NMOSD who are anti-aquaporin-4 Ab+
- The opinion was based on the P-III trial (CHAMPION-NMOSD) evaluating Ultomiris in 58 patients across North America, the EU, Asia-Pacific & Japan. The trial met its 1EPs of time to first on-trial relapse as confirmed by an independent adjudication committee & showed zero relapses with a median treatment duration of 73wks. (relapse risk reduction 98.6%) & continuing through a median duration of 90wks.
- The safety & tolerability were consistent with prior studies and real-world use with no new safety signals. The regulatory submissions for Ultomiris are under review with multiple health authorities, incl. in the US & Japan
Rinvoq
Active ingredient: upadacitinib Approved: April 17, 2023
Company: AbbVie Disease: Active Crohn's Disease
- The approval was based on 3 P-III trials incl. (U-EXCEED & U-EXCEL) induction studies & (U-ENDURE) maintenance study to evaluate upadacitinib (45mg, qd) as IT & (15/30mg, qd) as MT vs PBO in adults
- The results showed that more patients achieved the co-1EPs in the induction & maintenance study treated with Rinvoq 45mg @12wk. and (15 & 30mg) @52wks. demonstrated an endoscopic response (35% & 46% vs 4% & 13%) and (28% & 40% vs 7%); clinical remission (40% & 51% vs 14% & 22%) and (36% & 46% vs 14%), respectively
- In the 2EPs & additional EPs from the IT & MT, corticosteroid-free clinical remission (37% & 44% vs 7% & 13%) and (35% & 45% vs 14%); mucosal healing (17% & 25% vs 0% & 5%) and (13% & 24% vs 4%). The safety profile was consistent with the known safety profile of Rinvoq
Opzelura
Active ingredient: ruxolitinib Approved: April 21, 2023
Company: Incyte Disease: Non-Segmental Vitiligo
- The EC has granted marketing authorization for Opzelura (15mg/g) to treat adults & adolescents aged ≥12yrs. The EC decision was based on the P-III trials (TRuE-V1 & V2) evaluating Opzelura vs vehicle in 600+ patients aged ≥12yrs.
- The results showed an improvement in facial & total body repigmentation as shown by the no. of patients who reached F-VASI-T-VASI EPs @24wk. & in an OLE @52wk.
- ≥75% improvement from baseline in F-VASI75 in both studies (29.8% & 30.9% vs 7.4% & 11.4%), respectively while one in two patients achieved F-VASI75 @52wk. & one in three @52wk., ≥15% vs ~2% achieved ≥90% improvement in F-VASI (F-VASI90) @24wk., no serious TRAEs related to ruxolitinib were reported. The EC’s decision applies to all 27 EU Member States, Iceland, Norway & Liechtenstein
Akeega
Active ingredient: niraparib & abiraterone acetate Approved: April 21, 2023
Company: Janssen Disease: Metastatic Castration-Resistant Prostate Cancer
- The EC has granted marketing authorization for Akeega in the form of a dual-action tablet for mCRPC. The authorization was based on the P-III trial (MAGNITUDE) evaluating niraparib + AA & prednisone or prednisolone vs PBO + abiraterone acetate & prednisone in 765 patients
- The results showed an improvement in rPFS in all HRR+ patients with 47% risk reduction in patients with BRCA1/2 gene mutations. At the median follow-up at 24.8mos. in the BRCA subgroup, rPFS showed a consistent & clinical treatment effect with m-rPFS (19.5 vs 10.9mos.)
- A trend towards improved OS, improvement in TSP & clinical improvement in time-to-initiation of cytotoxic CT. The safety profile was consistent with the known safety profile of each agent, grade 3/4 AEs in patients with HRR gene alterations (67% vs 46.4%) with maintained overall QoL
Vafseo
Active ingredient: vadadustat Approved: April 26, 2023
Company: Akebia Disease: Symptomatic Anaemia
- The approval was based on the results from a comprehensive development program of ~7,500 patients, incl. the P-III (INNO2VATE) program of vadadustat (HIF-PH inhibitor) for the treatment of anemia due to CKD in adult patients on dialysis
- In each of the two (INNO2VATE) studies, vadadustat met the primary & secondary efficacy EPs which was found to be non-inferior to darbepoetin alfa as measured by a mean change in Hb b/w baseline & primary evaluation period (24-36wk.) and secondary evaluation period (40-52wk.)
- The therapy also achieved the primary safety EPs & was non-inferior to darbepoetin alfa in time to 1st occurrence of major adverse cardiovascular EPs. The approval is valid for all 27 EU member states, Iceland, Norway & Liechtenstein
Cosentyx
Active ingredient: secukinumab Approved: April 26, 2023
Company: Novartis Disease: Hidradenitis Suppurativa
- The EMA’s CHMP has adopted a positive opinion recommending marketing authorization of Cosentyx. The opinion was based on the P-III trials (SUNSHINE) & (SUNRISE) evaluating Cosentyx (300mg, q2w/q4w) vs PBO in 545 & 544 patients across 40 countries
- In both trials, patients treated with Cosentyx (300mg, q2w) achieved HiSCR (45.0% vs 33.7%) & (42.3% vs 31.2%) @16wks.; (46.1% vs 31.2%) & (41.8% vs 33.7%) with Cosentyx (300mg, q4w). Treatment response rates continued to improve beyond the 1EPs analysis @16wk.
- ≥55% achieved a HiSCR measure @52wk., ≥50% reduction in pain. The safety results were consistent with the well-established profile of Cosentyx & have been submitted to the US FDA with decisions expected in 2023
Opfolda
Active ingredient: miglustat Approved: April 26, 2023
Company: Amicus Therapeutics Disease: Late-Onset Pompe Disease
- The EMA’s CHMP has adopted a positive opinion recommending marketing authorization of miglustat. The EC’s decision & commercial launch of Pombiliti + Opfolda is expected in Q3’23
- The opinion was based on the P-III trial (PROPEL) evaluating the efficacy, safety & tolerability of Pombiliti. AT-GAA showed improvements in musculoskeletal & respiratory measures in clinical studies
- If Pombiliti + Opfolda is approved, it will be the first two-component therapy available in the EU for the treatment of adults with LOPD. Pombiliti + Opfolda also known as AT-GAA, the two-component investigational therapy that combines cipaglucosidase alfa to enable high-affinity uptake through the M6P receptor
Columvi
Active ingredient: glofitamab Approved: April 26, 2023
Company: Roche Disease: Diffuse Large B-Cell Lymphoma
- The EMA’s CHMP has recommended the approval of Columvi for adult patients with r/r DLBCL after 2 lines of systemic therapy. The EC’s final decision is expected in the near future
- The recommendation was based on the pivotal cohort in the P-I/II (NP30179) study evaluating Columvi. The results showed that Columvi given as a fixed course induced a CR (35.2%), ORR (50%) while 74.6% continued to experience a response @12mos. who achieved CR, the median duration of CR (not reached), and the median follow-up for DoR was 12.8mos. & median time to first CR was 42 days
- 1 patient discontinued treatment due to CRS while the NEJM provided additional data from a larger cohort in the (NP30179) study. Columvi was approved in Health Canada for r/r DLBCL
Camzyos
Active ingredient: mavacamten Approved: April 27, 2023
Company: BMS Disease: Obstructive Hypertrophic Cardiomyopathy
- The EMA’s CHMP has recommended approval of Camzyos (cardiac myosin inhibitor) for symptomatic (New York Heart Association, NYHA, class II-III) obstructive HCM
- The opinion was based on 2 P-III trials (EXPLORER-HCM) & (VALOR-HCM) evaluating Camzyos vs PBO in 251 & 112 adult patients. Both trials met their 1EPs & 2EPs & showed a clear treatment effect in the P-III trial (EXPLORER-HCM) with clinical improvements in exercise capacity, symptoms, and functional status along with an improvement in LVOTO
- The P-III trial (VALOR-HCM) showed an improvement across cardiac measures who met the 2011 ACC/AHA or 2014 ESC guideline criteria for SRT & were referred for an invasive procedure. The therapy also showed a reduction in need or eligibility for SRT
Orkambi
Active ingredient: umacaftor/ivacaftor Approved: April 27, 2023
Company: Vertex Disease: Cystic Fibrosis
- The EMA’s CHMP has adopted a positive opinion for the label extension of Orkambi (lumacaftor/ivacaftor) to treat children with CF aged 1-<2yrs. who have two copies of the F508del mutation in the CFTR gene
- The expanded approval was based on the P-III clinical trial evaluating Orkambi in children aged 1-<2yrs. which was found to be safe and well tolerated, and reduced the sweat chloride concentration
- Orkambi was approved in the EU for the treatment of patients with CF aged ≥2yrs. who have two copies of the F508del mutations. If Orkambi is approved, a treatment option will be available for the first time for about 300 young children with CF
Note: Breyanzi, Ultomiris, Cosentyx, Opfolda & Orkambi received EMA’s CHMP Positive Opinion & EC’s Marketing Authorization for Akeega, Vafseo while CHMP Recommendation for Approval of Columvi
Related Post: Insights+: EMA Marketing Authorization of New Drugs in March 2023

Neha is a Senior Editor at PharmaShots. She is passionate and very enthusiastic about recent updates and developments in the life sciences and pharma industry. She covers Biopharma, MedTech, and Digital health segments along with different reports at PharmaShots. She can be contacted at connect@pharmashots.com.














